BASF on Twitter: "With vinyl methyl oxazolidinone(VMOX)BASF offers new vinyl monomer for various applications.Particularly as reactive in UV curing inks+coatings,VMOX combines technical benefits as very low viscosity+very high reactivity with favorable ...

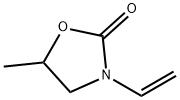
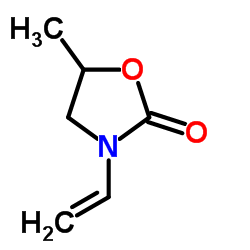
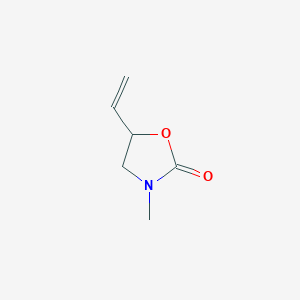
5-Methyl-3-vinyl-2-oxazolidinone–Investigations of a New Monomer for Kinetic Hydrate Inhibitor Polymers | Energy & Fuels

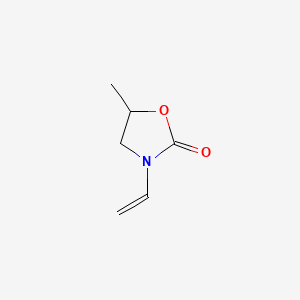
New BASF vinyl monomer for UV curing products in the printing and coatings industry – Paints and Coatings Expert
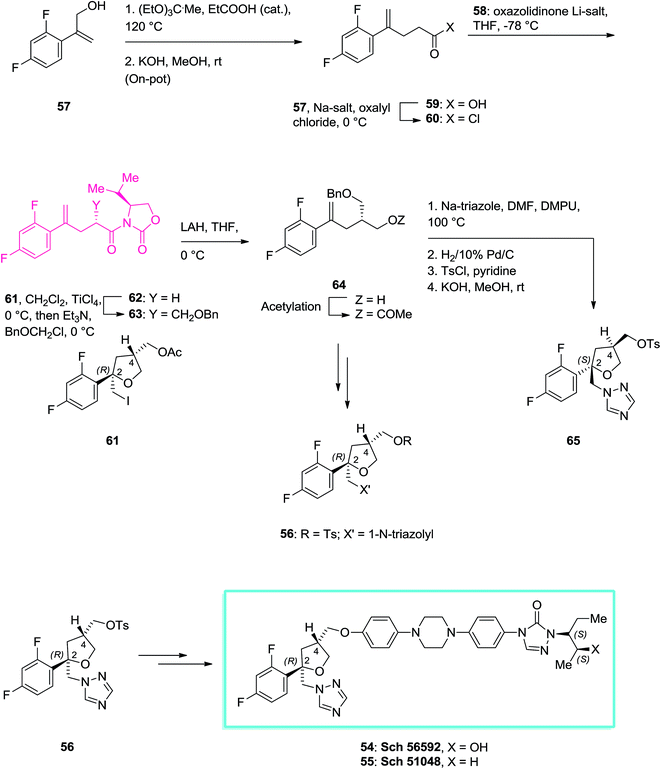
Applications of oxazolidinones as chiral auxiliaries in the asymmetric alkylation reaction applied to total synthesis - RSC Advances (RSC Publishing) DOI:10.1039/C6RA00653A

5-Methyl-3-vinyl-2-oxazolidinone–Investigations of a New Monomer for Kinetic Hydrate Inhibitor Polymers | Energy & Fuels
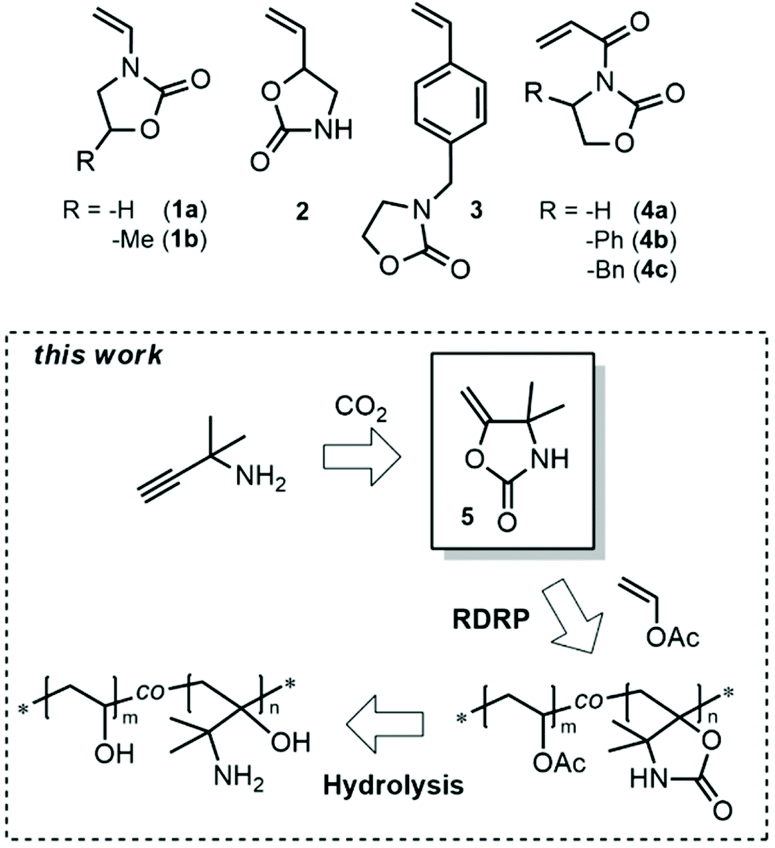
Reversible deactivation radical (co)polymerization of dimethyl methylene oxazolidinone towards responsive vicinal aminoalcohol-containing copolymers - Polymer Chemistry (RSC Publishing) DOI:10.1039/D0PY01255F
![3 + 1 + 1] cyclization of vinyl oxiranes with azides and CO by tandem palladium catalysis: efficient synthesis of oxazolidinones - Organic Chemistry Frontiers (RSC Publishing) DOI:10.1039/D1QO00591J 3 + 1 + 1] cyclization of vinyl oxiranes with azides and CO by tandem palladium catalysis: efficient synthesis of oxazolidinones - Organic Chemistry Frontiers (RSC Publishing) DOI:10.1039/D1QO00591J](https://pubs.rsc.org/image/article/2021/QO/d1qo00591j/d1qo00591j-s2_hi-res.gif)
3 + 1 + 1] cyclization of vinyl oxiranes with azides and CO by tandem palladium catalysis: efficient synthesis of oxazolidinones - Organic Chemistry Frontiers (RSC Publishing) DOI:10.1039/D1QO00591J

Lucia Veltri's research works | Università della Calabria, Rende (Università della Calabria) and other places
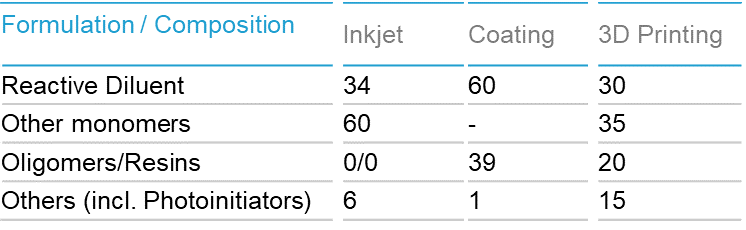
Reactive Diluents to Overcome Challenges in UV-Curable Inkjet Inks and Coatings Applications - UV+EB Technology
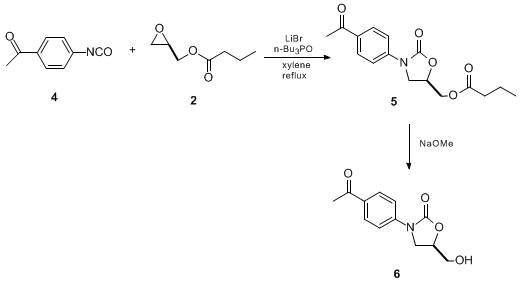
4-methyl-1,3-oxazolidin-2-one (16112-59-7) - Chemical Safety, Models, Suppliers, Regulation, and Patents - Chemchart

Recent Advances in the Synthesis and Ring‐Opening Transformations of 2‐ Oxazolidinones - Sun - 2021 - Advanced Synthesis & Catalysis - Wiley Online Library

Oxazolidinones as chiral auxiliaries in asymmetric aldol reaction applied to natural products total synthesis - ScienceDirect

TiCl4‐Promoted Asymmetric Aldol Reaction of Oxazolidinones and its Sulphur‐Congeners for Natural Product Synthesis - Bhamboo - 2021 - Asian Journal of Organic Chemistry - Wiley Online Library