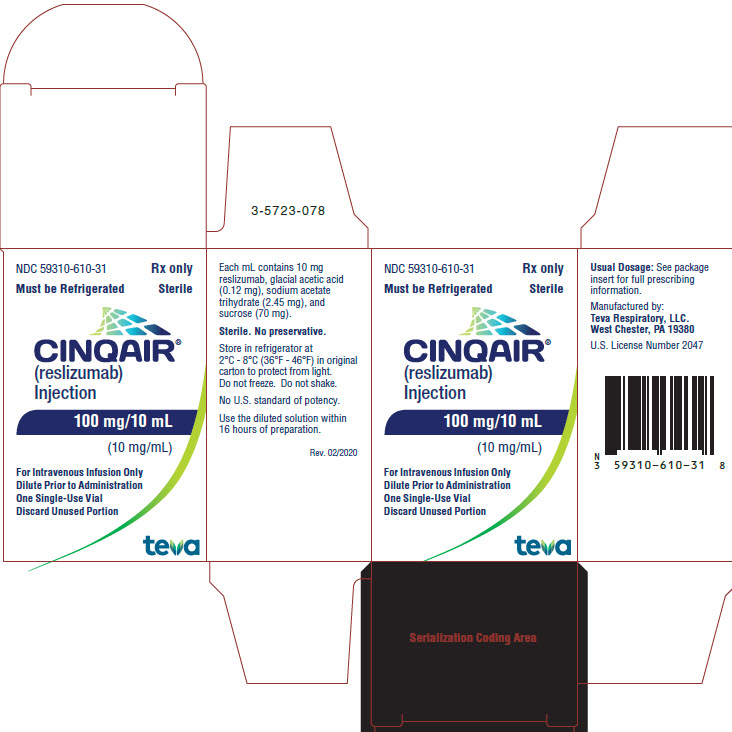
These highlights do not include all the information needed to use CINQAIR safely and effectively. See full prescribing information for CINQAIR.CINQAIR® (reslizumab) injection, for intravenous useInitial U.S. Approval: 2016

Cinqair (reslizumab) Injection for treatment of Severe Asthma and an Eosinophilic Phenotype - Clinical Trials Arena
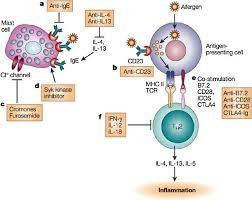
Predicting Responders to Reslizumab after 16 Weeks of Treatment Using an Algorithm Derived from Clinical Studies of Patients wit