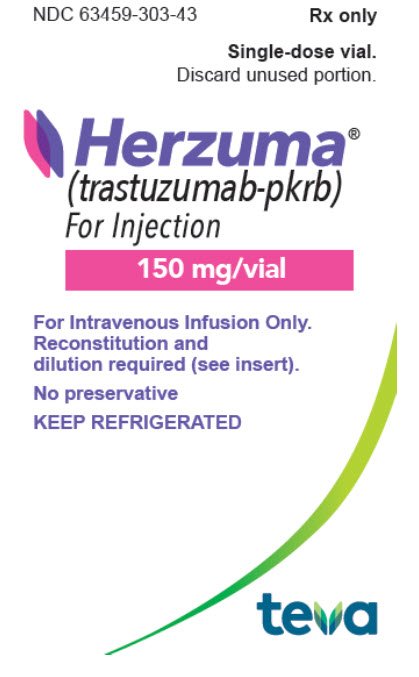
Teva and Celltrion Healthcare Announce U.S. Availability of HERZUMA® (trastuzumab-pkrb) for Injection | Business Wire

Celltrion and Teva Announce FDA Approval of Herzuma, a Biosimilar to Herceptin, for Treatment of HER2-Overexpressing Breast Cancer | Specialty Pharma Journal
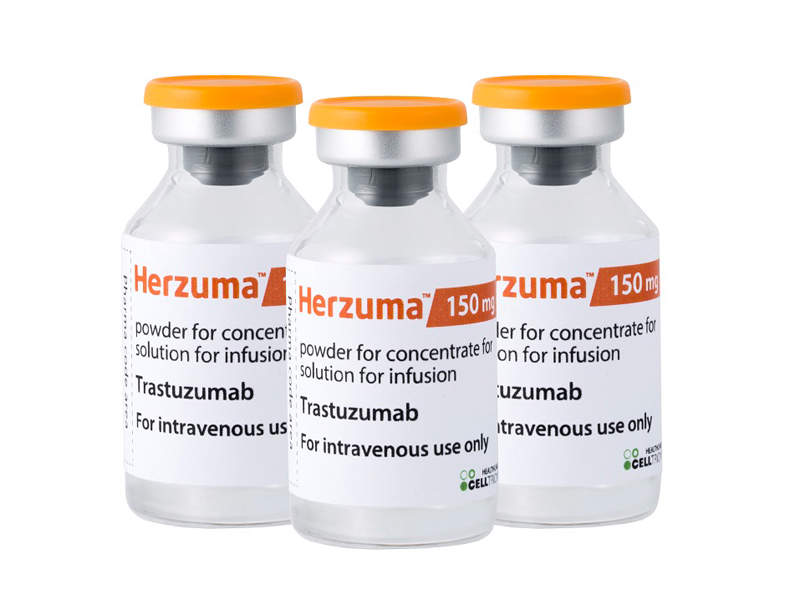
Herzuma (trastuzumab biosimilar) for the Treatment of Breast and Gastric Cancers - Clinical Trials Arena